Solutions to Key Technical Problems in the Manhattan Project
by
Ardill
The Manhattan Project may have been the greatest scientific collaboration of all time. It engaged, on a single project, a collection of the best minds in physics of that time, including notables such as Oppenheimer, Lawrence, Feynman, Fermi, Bohr, Bethe, Teller, von Neumann, Szilard, Compton, Chadwick, Rabi, Segre, Alvarez, Wheeler, and Wigner. Although the sheer scale of the project (the direct contribution of 600,000 Americans)[1] required a number of non-technical problems be solved, including management, procurement, safety, testing, logistics, and security, this paper focuses on the chief technical problems that needed to be solved to make a usable nuclear weapon.
Uranium Separation
This was among the first problems faced by the Manhattan Project. The most abundant isotope of uranium, namely U-238, is not usable for bomb-building, because it is not able to undergo a sustained nuclear fission reaction, as would be necessary for proper detonation. U-235, however, fissions “with high probability at very low neutron energy: ~0.01 to 0.1 eV.”[2] However, because U-235 makes up only 0.72% of naturally occuring uranium,[3] it is hard to get enough fissile material to make a bomb. “U-235 could be separated by means of a centrifuge, diffusion, and electromagnetic processes…. These processes appeared to be nearly ready for pilot plant construction.”[4] Later, a fourth process, liquid thermal diffusion, was added.[5]
Centrifuge
This technique involved adding uranium hexafluoride gas (UF6) of both isotopes to a centrifuge cylinder, and spinning it very fast. “This rotation creates a strong centrifugal force so that the heavier gas molecules (UF6 containing U-238 atoms) move towards the outside of the cylinder. The lighter gas molecules (containing U-235) collect closer to the center.”[6] This process is effective, (and was later used in the cold war) but it was abandoned because it was not making significant progress compared to other methods.[7]
Gaseous Diffusion
This solution was based upon the principle that gaseous molecules of lower mass move faster than gaseous molecules of higher mass when they are at the same temperature. This was exploited by passing UF6 gas through a membrane, allowing the molecules containing U-235 to become slightly more concentrated near the front of the gas cloud. By repeating this process hundreds of times, the producers were able to attain weapons-grade concentrations of U-235. However, this would fail if there was so much as a crack in a pipe: moisture from the air outside could react with the UF6 and cause some of it to solidify, clogging up the membrane. Also, UF6 corrodes steel, meaning that all piping had to be coated in nickel (a metal that UF6 does not react with) on the inside. This technique was used most famously at the K-25 plant in Oak Ridge, TN, which was created as a backup plant for Y-12. “Surprising many and relieving everyone, [it operated] almost flawlessly.”[8]
Electromagnetic separation
This relied on the fact that multiple charged particles moving through a magnetic field at the same inclination will be repelled with the same force. Because an equal force will push lighter particles farther than heavier ones, U-235 ions would be pushed farther than U-238 ions of the same charge. This separated a stream of uranium ions into two streams, one for the lighter particles and one for the heavier ones. Those went to separate (negatively charged) collectors, where they were converted back to non-ionic metal atoms. This was implemented at the Y-12 plant.[9]
One problem was that because the difference in mass was so small, the required power of the magnetic fields was colossal. As Theodore Rockwell, an engineer at Y-12, said, “If you walked along the wooden catwalk over the magnet you could feel the tug of the magnetic field on the nails in your shoes. It was like walking through glue.”[10] Watches that were brought too near to the magnet would have all of their internal components smashed by the force.
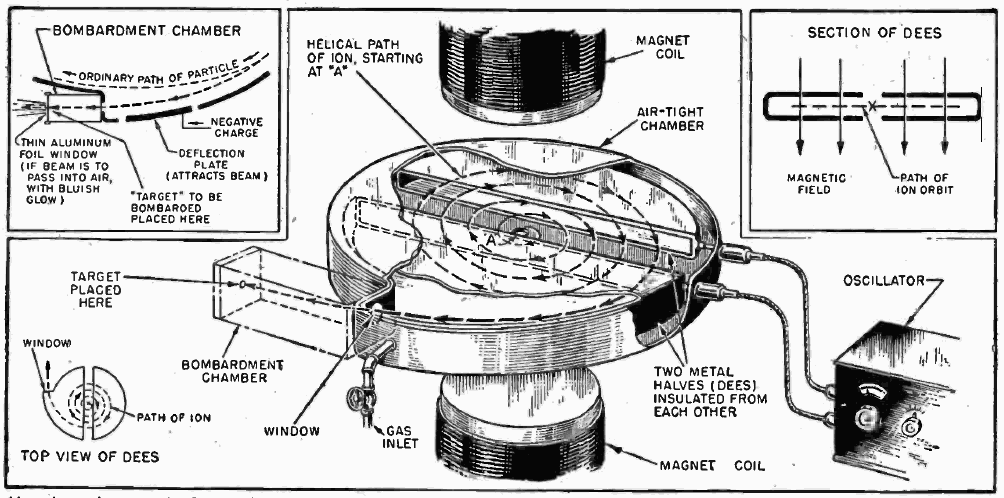
Another problem arose in making sure that all of the ions were at the same velocity. This was solved by using cyclotron technology - accelerating the charged particles by varying the electrical charge on two hollow D-shaped shells (with space in between) to alternatively pull and push a charged particle while placing it under the influence of a magnetic field, making it rotate in a spiral path inside the cyclotron vacuum chamber, and eventually shooting out through a hole in the side.[11] The cyclotron was the invention of Ernest Lawrence, one of the scientists working in the Manhattan Project.[12]
Uranium Separation
This was among the first problems faced by the Manhattan Project. The most abundant isotope of uranium, namely U-238, is not usable for bomb-building, because it is not able to undergo a sustained nuclear fission reaction, as would be necessary for proper detonation. U-235, however, fissions “with high probability at very low neutron energy: ~0.01 to 0.1 eV.”[2] However, because U-235 makes up only 0.72% of naturally occuring uranium,[3] it is hard to get enough fissile material to make a bomb. “U-235 could be separated by means of a centrifuge, diffusion, and electromagnetic processes…. These processes appeared to be nearly ready for pilot plant construction.”[4] Later, a fourth process, liquid thermal diffusion, was added.[5]
Centrifuge
This technique involved adding uranium hexafluoride gas (UF6) of both isotopes to a centrifuge cylinder, and spinning it very fast. “This rotation creates a strong centrifugal force so that the heavier gas molecules (UF6 containing U-238 atoms) move towards the outside of the cylinder. The lighter gas molecules (containing U-235) collect closer to the center.”[6] This process is effective, (and was later used in the cold war) but it was abandoned because it was not making significant progress compared to other methods.[7]
Gaseous Diffusion
This solution was based upon the principle that gaseous molecules of lower mass move faster than gaseous molecules of higher mass when they are at the same temperature. This was exploited by passing UF6 gas through a membrane, allowing the molecules containing U-235 to become slightly more concentrated near the front of the gas cloud. By repeating this process hundreds of times, the producers were able to attain weapons-grade concentrations of U-235. However, this would fail if there was so much as a crack in a pipe: moisture from the air outside could react with the UF6 and cause some of it to solidify, clogging up the membrane. Also, UF6 corrodes steel, meaning that all piping had to be coated in nickel (a metal that UF6 does not react with) on the inside. This technique was used most famously at the K-25 plant in Oak Ridge, TN, which was created as a backup plant for Y-12. “Surprising many and relieving everyone, [it operated] almost flawlessly.”[8]
Electromagnetic separation
This relied on the fact that multiple charged particles moving through a magnetic field at the same inclination will be repelled with the same force. Because an equal force will push lighter particles farther than heavier ones, U-235 ions would be pushed farther than U-238 ions of the same charge. This separated a stream of uranium ions into two streams, one for the lighter particles and one for the heavier ones. Those went to separate (negatively charged) collectors, where they were converted back to non-ionic metal atoms. This was implemented at the Y-12 plant.[9]
One problem was that because the difference in mass was so small, the required power of the magnetic fields was colossal. As Theodore Rockwell, an engineer at Y-12, said, “If you walked along the wooden catwalk over the magnet you could feel the tug of the magnetic field on the nails in your shoes. It was like walking through glue.”[10] Watches that were brought too near to the magnet would have all of their internal components smashed by the force.
Another problem arose in making sure that all of the ions were at the same velocity. This was solved by using cyclotron technology - accelerating the charged particles by varying the electrical charge on two hollow D-shaped shells (with space in between) to alternatively pull and push a charged particle while placing it under the influence of a magnetic field, making it rotate in a spiral path inside the cyclotron vacuum chamber, and eventually shooting out through a hole in the side.[11] The cyclotron was the invention of Ernest Lawrence, one of the scientists working in the Manhattan Project.[12]
[13]
Liquid Thermal Diffusion
This was based on the theory that when a mixture of gases flows through a container that has a hot wall and a cold wall, the lighter of the two gases tends to gather near the hot wall. This is because molecules of both gases will heat up when they come in contact with the hot wall, gaining kinetic energy. They will then begin to hit the molecules surrounding them. The lighter molecules, when hitting other molecules, will be more likely to bounce back toward the hot wall, while the heavier ones will be more likely to continue through toward the cold one. This will increase the concentration of lighter molecules near the hot wall steadily over time.
This was applied by pumping UF6 through the space between two vertical concentric pipes. The inner pipe was filled with steam, and the outer one was surrounded by cold water. The outer pipe was the cold wall, and the inner one was the hot wall. Because the the U(-235)F6 molecules were slightly lighter than their U-238 counterparts, U(-235)F6 gathered near the hot wall, while U(-238)F6 gathered near the cold one. The warmer U(-235)F6 then rose above the the colder U(-238)F6 because of the convection current, and it was
collected at the top.
[Images pending permission for use]
[14][15]
This process was used as a feeder for gaseous diffusion and electromagnetic separation, which were very power-intensive, and were not turning out high enough concentrations of U-235 to be useful. The liquid thermal diffusion plant (S-50) “was the first stage of uranium enrichment, from 0.72% to 0.85% U-235”,[16] the output from which was then reprocessed in the other plants.
Detonation
This was the most important problem that the Manhattan Project had to solve. It was the problem of exploding a bomb at the right time, not before or after. Essentially, to explode, the bomb must assemble a critical mass of the metal, or reflect enough neutrons onto the metal to simulate the effect of a critical mass. An important note is that when an atom is struck by a neutron, it does not necessarily undergo fission. It may have a high probability, but it is not definite. This is why it is crucial to have thousands of atoms undergoing fission to be sure of a sustained reaction. Also, decay of the metal itself does not always supply the needed neutrons, which is why some designs incorporate a polonium-beryllium starter. Of the designs proposed, one used a sphere of beryllium to reflect neutrons, another was the famous “gun barrel” design used in Little Boy, and the third is the plutonium implosion design. Only the last two were used.
Beryllium Sphere
This was based off of the idea that a critical mass can be simulated at a subcritical mass by reflecting all neutrons that are emitted back onto the chunk of metal, so that the neutrons that would normally be lost into the air are instead reflected back, allowing more atoms to be struck by neutrons, and therefore increasing the chance of detonation dramatically. This design consisted of a hollow beryllium sphere filled with uranium, with a plug removed, as shown in the image below.[17] The ball in the center is a polonium-beryllium starter, which was in the design just in case the uranium did not go off by itself. The idea was “to use a sphere and to shoot into it a cylindrical plug made of some [fissile] material and some [neutron reflector].”[18]
[Image pending permission for use]
[19]
This method was not pursued because it required that the expansion “proceed only inward”[20] if the bomb was to have a high efficiency. “Of course in actual fact it will proceed outward as well as inward and outward expansion would in reality give the dominant effect.”[21] Even if the outward expansion were small, the method would be very inefficient: “an efficiency of only about 10–9 was calculated.”[22]
Gun-setup
This was perhaps the most elementary of the proposed designs, which may have contributed to its success. It worked by keeping two subcritical masses of uranium separate until the moment of detonation, at which point a high explosive charge would send one colliding into the other. This would create a critical mass, and therefore cause a nuclear explosion.
[Image pending permission for use]
[23]
This setup was proposed for use with both uranium and plutonium, but it was only used with uranium, because the creation process for plutonium ended with two isotopes: Pu-239 and Pu-240. Pu-239 was normal, fissile material, but Pu-240 was able to undergo spontaneous fission. This means that it would trigger the bomb prematurely, causing the bomb to “fizzle out” instead of doing maximum damage.[24] (To be exact, % of a given sample of Pu-240 will undergo spontaneous fission, but because of how many plutonium atoms were involved, that could yield a rather large effect.)[25]
Plutonium Implosion
This was created as a substitute for the gun setup, because a plutonium gun setup had reliability issues. It used explosive shaped charges to quickly compress a subcritical hollow shell of a sphere made from plutonium equally from all sides. The speed was an important factor here, because the compression had to be faster than Pu-240’s spontaneous fission, if they wished to prevent the fizzling that would occur in a plutonium gun setup. Also, it was crucial that the force be equal, so that the plutonium would reach a critical density: a density at which the neutrons inside the plutonium (from decay) would be nearly guaranteed to hit a nucleus, rather than having a possibility of going straight through the electron orbitals of the atoms it encounters. (For reference, a uranium atom is times as large as its nucleus.)[26] The compression would increase the chances of sustained fission significantly, making the bomb very likely to go off.
Because there was still a chance of it not going off, a polonium-beryllium starter was placed in the middle of the hollow plutonium sphere. It was made to fire neutrons at the surrounding plutonium at the moment of compression, so that there would be a much higher chance of detonation, because of the extra neutrons in the system.
Plutonium Production
Plutonium, an element that is much more fissile than uranium, was needed for the second bomb. It was produced through the decay of U-239 to Np-239 by means of β- decay. This has a half-life of 23.45 minutes.[27] Np-239 then decays to Pu-239 through β- decay with a half-life of 2.356 days.[28] Because U-239 does not occur naturally, it must be created by neutron absorption from U-238, a process which is far more likely to happen for low-energy neutrons. There were two designs to slow down the neutrons: “the uranium-graphite pile [and] the uranium-heavy-water pile”[29]
Uranium-Graphite Pile
This design involved sliding graphite control rods in and out of the reactor to moderate the number and speed of neutrons. In “the reactor, [neutrons from fission] are slowed down in a graphite (carbon) and some of them are captured in U-238 to produce the isotope U-239.”[30] Graphite “safety rods were suspended above the pile so that, in an emergency, they could be instantaneously released… and, as a last resort, arrangements were made to flood the pile with moderating chemicals.”[31] If the chemicals “had to be employed, the pile would no longer be usable.”[32] Both piles also incorporated a waste disposal system: a long array of holding tanks in which the cooling water was decontaminated. “The cooling water leaving the pile [was expected] to contain radioactive materials with relatively short half-lives…. The design provided for conducting this water underground to a retention basin for its final radioactive decay.”[33] This was done “to avoid any possibility of injury to fish in the Columbia River.”[34]
Uranium-Heavy-Water Pile
This design was very much like the last, but it used heavy water (water made from hydrogen-2 rather than hydrogen-1) to slow down neutrons. Heavy water’s lower rate of neutron absorption gave it a distinct advantage over a light water pile,[35] because there was a lower chance of tritium (a radioactive hydrogen isotope) creation. This reduced contamination.
This project stimulated huge advancements in the field of nuclear physics, while also decisively ending WWII. It required enormous investment, both monetary and temporal, by thousands of Americans, and it changed the world forever. Many of the technical challenges that it traversed were only solved due to the greatest accumulation of talent in the twentieth century. Even the greatest scientists have ideas that fail, but what differentiates them from all others is their ability to have a secondary plan, every time. This project is a testament to their ingenuity, and it stands forever as one of science’s greatest achievements.
Primary Sources
Secondary Sources
Glossary
Footnotes
[1] Leslie R. Groves, Now It Can Be Told: The Story of the Manhattan Project (New York, NY: Da Capo Press, 1975), 414.
[2] "Nuclear Reactors," University of Washington Web Server, , accessed January 21, 2019, http://depts.washington.edu/jrphys/ph248S16/reactors1.pdf.
[3] Patricia Shapely, "Fuel for Fission," , accessed January 22, 2019, http://butane.chem.uiuc.edu/pshapley/GenChem1/L7/2.html.
[4] Now It Can Be Told, 10.
[5] Ibid., 120.
[6] "Uranium Enrichment," United States Nuclear Regulatory Commission - Protecting People and the Environment, , accessed January 22, 2019, https://www.nrc.gov/materials/fuel-cycle-fac/ur-enrichment.html#centrifuge.
[7] Michael B. Stoff, The Manhattan Project: A Documentary Introduction to the Atomic Age (New York, NY: Black Dog & Leventhal Publishers, 2007), 74.
[8] Ibid., 199.
[9] Now It Can Be Told, 95.
[10] The Manhattan Project, 207.
[11] "Cyclotron," Total Internal Reflection, , accessed January 22, 2019, http://hyperphysics.phy-astr.gsu.edu/hbase/magnetic/cyclot.html.
[12] "Ernest O. Lawrence," Atomic Heritage Foundation, , accessed January 22, 2019, https://www.atomicheritage.org/profile/ernest-o-lawrence.
[13] Source: https://upload.wikimedia.org/wikipedia/commons/f/fe/Cyclotron_diagram.png
[14] Source: https://cdn.iopscience.com/images/books/978-1-6270-5991-6/live/bk978-1-6270-5991-6ch3f14_online.jpg
[15] Source: https://www.osti.gov/opennet/manhattan-project-history/images/ThermalDiffusion1.jpg
[16] Cameron Reed, "Liquid Thermal Diffusion during the Manhattan Project," SAO/NASA ADS: ADS Home Page, June 01, 2011, , accessed January 22, 2019, http://adsabs.harvard.edu/abs/2011PhP....13..161C.
[17] A more precise design can be found at https://fas.org/sgp/othergov/doe/lanl/lib-www/la-pubs/00403725.pdf, pg. 9.
[18] Robert R. Serber, The Los Alamos Primer (Berkeley, CA: University of California Press, 1992), 56.
[19] Ibid., 61.
[20] Ibid.
[21] Ibid.
[22] Ibid., 62.
[23] Source: https://tse3.mm.bing.net/th?id=OIP.UU5sOGQjQyOW7hLNdrtZeQEsDF&pid=Api&w=273&h=181
[24] The Los Alamos Primer, 55.
[25] G. Audi et al., "The NUBASE2016 Evaluation of Nuclear Properties," Atomic Mass Data Center, March 10, 2017, 125, accessed January 21, 2019, http://amdc.in2p3.fr/nubase/2017Audi03.pdf.
[26] Based off of a 15 fm nuclear radius and a 230 pm atomic radius.
[27] "The NUBASE2016 Evaluation of Nuclear Properties," 125.
[28] Ibid.
[29] Now It Can Be Told, 10.
[30] The Los Alamos Primer, 14.
[31] Ibid., 88.
[32] Ibid.
[33] Ibid., 82.
[34] Ibid.
[35] Heavy water has a neutron cross section of ~17 barnes, vs light water’s ~100 barnes. Source:
Emmanual Farhi, "Light and Heavy Water Dynamic Structure Factor for Neutron Transport Codes," Taylor and Francis Online, , accessed January 21, 2019, https://www.tandfonline.com/doi/full/10.1080/00223131.2014.984002#_i18.
Liquid Thermal Diffusion
This was based on the theory that when a mixture of gases flows through a container that has a hot wall and a cold wall, the lighter of the two gases tends to gather near the hot wall. This is because molecules of both gases will heat up when they come in contact with the hot wall, gaining kinetic energy. They will then begin to hit the molecules surrounding them. The lighter molecules, when hitting other molecules, will be more likely to bounce back toward the hot wall, while the heavier ones will be more likely to continue through toward the cold one. This will increase the concentration of lighter molecules near the hot wall steadily over time.
This was applied by pumping UF6 through the space between two vertical concentric pipes. The inner pipe was filled with steam, and the outer one was surrounded by cold water. The outer pipe was the cold wall, and the inner one was the hot wall. Because the the U(-235)F6 molecules were slightly lighter than their U-238 counterparts, U(-235)F6 gathered near the hot wall, while U(-238)F6 gathered near the cold one. The warmer U(-235)F6 then rose above the the colder U(-238)F6 because of the convection current, and it was
collected at the top.
[Images pending permission for use]
[14][15]
This process was used as a feeder for gaseous diffusion and electromagnetic separation, which were very power-intensive, and were not turning out high enough concentrations of U-235 to be useful. The liquid thermal diffusion plant (S-50) “was the first stage of uranium enrichment, from 0.72% to 0.85% U-235”,[16] the output from which was then reprocessed in the other plants.
Detonation
This was the most important problem that the Manhattan Project had to solve. It was the problem of exploding a bomb at the right time, not before or after. Essentially, to explode, the bomb must assemble a critical mass of the metal, or reflect enough neutrons onto the metal to simulate the effect of a critical mass. An important note is that when an atom is struck by a neutron, it does not necessarily undergo fission. It may have a high probability, but it is not definite. This is why it is crucial to have thousands of atoms undergoing fission to be sure of a sustained reaction. Also, decay of the metal itself does not always supply the needed neutrons, which is why some designs incorporate a polonium-beryllium starter. Of the designs proposed, one used a sphere of beryllium to reflect neutrons, another was the famous “gun barrel” design used in Little Boy, and the third is the plutonium implosion design. Only the last two were used.
Beryllium Sphere
This was based off of the idea that a critical mass can be simulated at a subcritical mass by reflecting all neutrons that are emitted back onto the chunk of metal, so that the neutrons that would normally be lost into the air are instead reflected back, allowing more atoms to be struck by neutrons, and therefore increasing the chance of detonation dramatically. This design consisted of a hollow beryllium sphere filled with uranium, with a plug removed, as shown in the image below.[17] The ball in the center is a polonium-beryllium starter, which was in the design just in case the uranium did not go off by itself. The idea was “to use a sphere and to shoot into it a cylindrical plug made of some [fissile] material and some [neutron reflector].”[18]
[Image pending permission for use]
[19]
This method was not pursued because it required that the expansion “proceed only inward”[20] if the bomb was to have a high efficiency. “Of course in actual fact it will proceed outward as well as inward and outward expansion would in reality give the dominant effect.”[21] Even if the outward expansion were small, the method would be very inefficient: “an efficiency of only about 10–9 was calculated.”[22]
Gun-setup
This was perhaps the most elementary of the proposed designs, which may have contributed to its success. It worked by keeping two subcritical masses of uranium separate until the moment of detonation, at which point a high explosive charge would send one colliding into the other. This would create a critical mass, and therefore cause a nuclear explosion.
[Image pending permission for use]
[23]
This setup was proposed for use with both uranium and plutonium, but it was only used with uranium, because the creation process for plutonium ended with two isotopes: Pu-239 and Pu-240. Pu-239 was normal, fissile material, but Pu-240 was able to undergo spontaneous fission. This means that it would trigger the bomb prematurely, causing the bomb to “fizzle out” instead of doing maximum damage.[24] (To be exact, % of a given sample of Pu-240 will undergo spontaneous fission, but because of how many plutonium atoms were involved, that could yield a rather large effect.)[25]
Plutonium Implosion
This was created as a substitute for the gun setup, because a plutonium gun setup had reliability issues. It used explosive shaped charges to quickly compress a subcritical hollow shell of a sphere made from plutonium equally from all sides. The speed was an important factor here, because the compression had to be faster than Pu-240’s spontaneous fission, if they wished to prevent the fizzling that would occur in a plutonium gun setup. Also, it was crucial that the force be equal, so that the plutonium would reach a critical density: a density at which the neutrons inside the plutonium (from decay) would be nearly guaranteed to hit a nucleus, rather than having a possibility of going straight through the electron orbitals of the atoms it encounters. (For reference, a uranium atom is times as large as its nucleus.)[26] The compression would increase the chances of sustained fission significantly, making the bomb very likely to go off.
Because there was still a chance of it not going off, a polonium-beryllium starter was placed in the middle of the hollow plutonium sphere. It was made to fire neutrons at the surrounding plutonium at the moment of compression, so that there would be a much higher chance of detonation, because of the extra neutrons in the system.
Plutonium Production
Plutonium, an element that is much more fissile than uranium, was needed for the second bomb. It was produced through the decay of U-239 to Np-239 by means of β- decay. This has a half-life of 23.45 minutes.[27] Np-239 then decays to Pu-239 through β- decay with a half-life of 2.356 days.[28] Because U-239 does not occur naturally, it must be created by neutron absorption from U-238, a process which is far more likely to happen for low-energy neutrons. There were two designs to slow down the neutrons: “the uranium-graphite pile [and] the uranium-heavy-water pile”[29]
Uranium-Graphite Pile
This design involved sliding graphite control rods in and out of the reactor to moderate the number and speed of neutrons. In “the reactor, [neutrons from fission] are slowed down in a graphite (carbon) and some of them are captured in U-238 to produce the isotope U-239.”[30] Graphite “safety rods were suspended above the pile so that, in an emergency, they could be instantaneously released… and, as a last resort, arrangements were made to flood the pile with moderating chemicals.”[31] If the chemicals “had to be employed, the pile would no longer be usable.”[32] Both piles also incorporated a waste disposal system: a long array of holding tanks in which the cooling water was decontaminated. “The cooling water leaving the pile [was expected] to contain radioactive materials with relatively short half-lives…. The design provided for conducting this water underground to a retention basin for its final radioactive decay.”[33] This was done “to avoid any possibility of injury to fish in the Columbia River.”[34]
Uranium-Heavy-Water Pile
This design was very much like the last, but it used heavy water (water made from hydrogen-2 rather than hydrogen-1) to slow down neutrons. Heavy water’s lower rate of neutron absorption gave it a distinct advantage over a light water pile,[35] because there was a lower chance of tritium (a radioactive hydrogen isotope) creation. This reduced contamination.
This project stimulated huge advancements in the field of nuclear physics, while also decisively ending WWII. It required enormous investment, both monetary and temporal, by thousands of Americans, and it changed the world forever. Many of the technical challenges that it traversed were only solved due to the greatest accumulation of talent in the twentieth century. Even the greatest scientists have ideas that fail, but what differentiates them from all others is their ability to have a secondary plan, every time. This project is a testament to their ingenuity, and it stands forever as one of science’s greatest achievements.
Primary Sources
- Serber, Robert. The Los Alamos Primer. Berkeley, CA: University of California Press, 1992.
- Groves, Leslie R. Now It Can Be Told: The Story of the Manhattan Project. New York, NY: Da Capo Press, 1975.
- Fermi, Rachel, and Esther Samara. Picturing The Bomb: Photographs From the Secret World of the Manhattan Project. New York, NY: Harry N. Abrams Inc. Publishing, 1995.
- Seaborg, Prof. Glenn T. The Plutonium Story: The Journals of Professor Glenn T. Seaborg, 1939-1946. Edited by Ronald Kathren, Jerry Gough, and Gary Benefiel. Columbus, OH: Battelle Press, 1994.
Secondary Sources
- Audi, G., F. G. Condev, Meng Wang, W. J. Huang, and S. Naimi. "The NUBASE2016 Evaluation of Nuclear Properties." Atomic Mass Data Center. March 10, 2017. Accessed January 22, 2019. http://amdc.in2p3.fr/nubase/2017Audi03.pdf.
- "Cyclotron." Total Internal Reflection. Accessed January 22, 2019. http://hyperphysics.phy-astr.gsu.edu/hbase/magnetic/cyclot.html.
- "Ernest O. Lawrence." Atomic Heritage Foundation. Accessed January 22, 2019. https://www.atomicheritage.org/profile/ernest-o-lawrence.
- Farhi, Emmanuel. "Light and Heavy Water Dynamic Structure Factor for Neutron Transport Codes." Taylor and Francis Online. Accessed January 22, 2019. https://www.tandfonline.com/doi/full/10.1080/00223131.2014.984002#_i18.
- Note: The next three sources contain reprinted primary sources, often in their entirety.
- Kelly, Cynthia C. (editor). Remembering the Manhattan Project. Toh Tuck Link, Singapore: World Scientific Publishing Co. Pte. Ltd, 2004.
- Kelly, Cynthia C. (editor). The Manhattan Project: The Birth of the Atomic Bomb in the Words of Its Creators, Eyewitnesses, and Historians. New York, NY: Black Dog & Leventhal Publishers, Inc, 2007.
- Stoff, Michael B., Jonathan F. Fanton, and R. Hal Williams (editors). The Manhattan Project: A Documentary Introduction to the Atomic Age. Philadelphia, PA: Temple University Press, 1991.
- "Nuclear Reactors." University of Washington Web Server. Accessed January 21, 2019. http://depts.washington.edu/jrphys/ph248S16/reactors1.pdf.
- Reed, Cameron. "Liquid Thermal Diffusion during the Manhattan Project." SAO/NASA ADS: ADS Home Page. June 01, 2011. Accessed January 22, 2019. http://adsabs.harvard.edu/abs/2011PhP....13..161C.
- Shapely, Patricia. "Fuel for Fission." Accessed January 22, 2019. http://butane.chem.uiuc.edu/pshapley/GenChem1/L7/2.html.
- "Uranium Enrichment." United States Nuclear Regulatory Commission - Protecting People and the Environment. Accessed January 22, 2019. https://www.nrc.gov/materials/fuel-cycle-fac/ur-enrichment.html#centrifuge.
Glossary
- Atoms - Tiny particles that make up all of the universe. They are composed of a nucleus, in the center of the atom, and electrons that orbit around the nucleus. The nucleus,which is made up of protons and neutrons, constitutes essentially all of the mass of an atom.
- Electron - A negatively charged fundamental particle.
- Proton - A positively charged fundamental particle found in the nuclei of atoms. The number of protons in the nucleus of an atom determines which element that atom belongs to. A proton has mass 1 amu.
- Neutron - An uncharged fundamental particle. The number of neutrons determines which isotope an atom belongs to. A neutron has mass 1 amu. Free neutrons (ones that are not in nuclei) have energies determined by their speed. The energy is normally measured in eV, keV, or MeV.
- Isotope - An atom that has a specific number of protons and a specific number of neutrons. The naming system for an isotope is simple: Element-atomic mass. Atomic mass is calculated by adding the number of protons and neutrons. For example, Uranium-235 is an isotope of Uranium which has 92 protons (by nature of being Uranium) and 143 neutrons (this is specific to isotope 235). The atomic mass is 92+143=235, so the name is Uranium-235. Different isotopes can have different properties.
- eV - Electron volt. A unit of energy, equal to the amount of work done on an electron when moving it through a potential difference of 1 volt. Common variations include keV (thousand electron volts) and MeV (million electron volts).
- Decay - The act of an atom changing its state without any stimulation. It occurs due to unstable nuclei. In β+ decay, a neutron splits into a proton and an electron. In β- decay, a proton splits into a neutron and a positron (anti-electron). In ɑ decay, a helium nucleus (two protons, two neutrons) is emitted.
- Ion - An atom that has lost or gained electrons, meaning it has a positive or negative charge. In this paper, there are only references to metallic ions, which result from the loss of electrons, meaning that they acquire a positive charge.
- U/Uranium - An element with 92 protons. It is generally abbreviated U. It naturally occurs in three isotopes: U-238, U-235, and U-234. U-234 makes up only 0.0055% of raw uranium (compared to U-235’s 0.72% and U-238’s 99.2745%), so it can be discounted as negligible.
- U-235 - The fissile isotope of uranium, used in the “Little Boy” bomb.
- Fission - When an atom’s nucleus splits due to instability, releasing large amounts of power. Requires stimulation by a “starter force” such as a neutron. Normally emits multiple neutrons.
- Spontaneous Fission - When an atom undergoes fission, without a starter force. Notable examples of spontaneous fission are in Pu-240.
- Critical mass - The mass at which there are enough neutrons “bouncing around” inside a piece of metal (from decay) that every fission reaction will supply the neutrons to start multiple more.
- Half-life - The amount of time that will pass before half of a sample of a substance has decayed. For example, the half-life for U-239 → Np-239 is 23.45 minutes, so after 23.45 minutes, half of the uranium-239 in any given sample will remain, the rest having decayed to Np-239. After 46.90 minutes, ¼ of the original sample will remain, and so on.
- Np/Neptunium - An element with 93 protons. Decays to Pu-239.
- Pu/Plutonium - An element with 94 protons. The isotope used was Pu-239, but it was contaminated by Pu-240.
Footnotes
[1] Leslie R. Groves, Now It Can Be Told: The Story of the Manhattan Project (New York, NY: Da Capo Press, 1975), 414.
[2] "Nuclear Reactors," University of Washington Web Server, , accessed January 21, 2019, http://depts.washington.edu/jrphys/ph248S16/reactors1.pdf.
[3] Patricia Shapely, "Fuel for Fission," , accessed January 22, 2019, http://butane.chem.uiuc.edu/pshapley/GenChem1/L7/2.html.
[4] Now It Can Be Told, 10.
[5] Ibid., 120.
[6] "Uranium Enrichment," United States Nuclear Regulatory Commission - Protecting People and the Environment, , accessed January 22, 2019, https://www.nrc.gov/materials/fuel-cycle-fac/ur-enrichment.html#centrifuge.
[7] Michael B. Stoff, The Manhattan Project: A Documentary Introduction to the Atomic Age (New York, NY: Black Dog & Leventhal Publishers, 2007), 74.
[8] Ibid., 199.
[9] Now It Can Be Told, 95.
[10] The Manhattan Project, 207.
[11] "Cyclotron," Total Internal Reflection, , accessed January 22, 2019, http://hyperphysics.phy-astr.gsu.edu/hbase/magnetic/cyclot.html.
[12] "Ernest O. Lawrence," Atomic Heritage Foundation, , accessed January 22, 2019, https://www.atomicheritage.org/profile/ernest-o-lawrence.
[13] Source: https://upload.wikimedia.org/wikipedia/commons/f/fe/Cyclotron_diagram.png
[14] Source: https://cdn.iopscience.com/images/books/978-1-6270-5991-6/live/bk978-1-6270-5991-6ch3f14_online.jpg
[15] Source: https://www.osti.gov/opennet/manhattan-project-history/images/ThermalDiffusion1.jpg
[16] Cameron Reed, "Liquid Thermal Diffusion during the Manhattan Project," SAO/NASA ADS: ADS Home Page, June 01, 2011, , accessed January 22, 2019, http://adsabs.harvard.edu/abs/2011PhP....13..161C.
[17] A more precise design can be found at https://fas.org/sgp/othergov/doe/lanl/lib-www/la-pubs/00403725.pdf, pg. 9.
[18] Robert R. Serber, The Los Alamos Primer (Berkeley, CA: University of California Press, 1992), 56.
[19] Ibid., 61.
[20] Ibid.
[21] Ibid.
[22] Ibid., 62.
[23] Source: https://tse3.mm.bing.net/th?id=OIP.UU5sOGQjQyOW7hLNdrtZeQEsDF&pid=Api&w=273&h=181
[24] The Los Alamos Primer, 55.
[25] G. Audi et al., "The NUBASE2016 Evaluation of Nuclear Properties," Atomic Mass Data Center, March 10, 2017, 125, accessed January 21, 2019, http://amdc.in2p3.fr/nubase/2017Audi03.pdf.
[26] Based off of a 15 fm nuclear radius and a 230 pm atomic radius.
[27] "The NUBASE2016 Evaluation of Nuclear Properties," 125.
[28] Ibid.
[29] Now It Can Be Told, 10.
[30] The Los Alamos Primer, 14.
[31] Ibid., 88.
[32] Ibid.
[33] Ibid., 82.
[34] Ibid.
[35] Heavy water has a neutron cross section of ~17 barnes, vs light water’s ~100 barnes. Source:
Emmanual Farhi, "Light and Heavy Water Dynamic Structure Factor for Neutron Transport Codes," Taylor and Francis Online, , accessed January 21, 2019, https://www.tandfonline.com/doi/full/10.1080/00223131.2014.984002#_i18.